Complete Turn-Key Solution
- Instruments
- Kits
- Training
- Service
- Support

The process
1. Install the kit
User scans the QR code on the kit to load the correct analysis program and installs the consumables from Tracer-QC kit onto the Tracer-QC instrument.

Tracer-QC confirms that the kit was installed properly, executes automated self-checks and indicates that it is ready to receive the sample.
Tracer-QC robot mixes the sample with indicators and dispenses the mixtures into the analysis plate. The plate reader analyses the optical signals (absorbed or emitted light) in the analysis plate.
2. Deliver the sample

The sample vial provided in the kit is placed inside a special pig that provides shielding and is designed to mate to Tracer-QC. QC sample is added to the vial in the pig from a QC syringe inside a hot cell. The pig is then carried to Tracer-QC and placed on the deck behind shielding where it is accessible by the pipetting robot.
3. Initiate the analysis
User initiates the analysis using the software

Tracer-QC robot mixes the sample with indicators and dispenses the mixtures into the analysis plate. The plate reader analyses the optical signals (absorbed or emitted light) in the analysis plate.

The software then analyses their optical characteristics against a set of predefined and validated reference specifications and translates them into the required QC measurements.
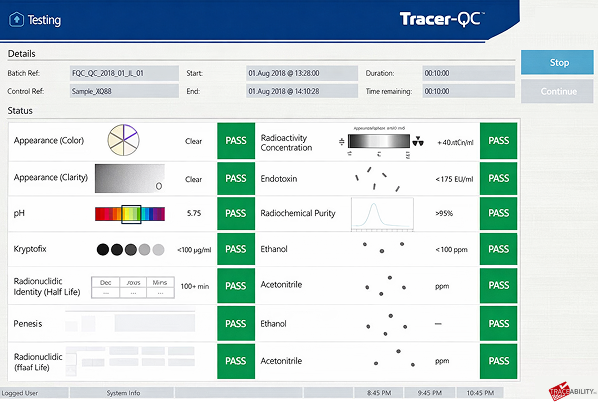
4. Collect the report
A single page summary is delivered automatically with all QC values and pass/fail determination supported by a detailed 26-page report with all measurements of sample and standard obtained during the process as well as suitability test data.

Features & Benefits
Ease of Compliance
No room for missing a process, record or signature
- Objective measurements with reference to standards and without any human interaction
- Data flow from measurement to batch record
— Automated
— Uncompromised
— Completely traceable
- Regular automated suitability testing with permanent record
- 21 CFR 11 compliant software with access control
- No cross-contamination samples never leave disposable kit (except rHPLC))
- Ease of audit (internal or FDA) – instantaneous data retrieval
Efficiency and Safety
- 1 electronic report with all QC results, automatically generated
- Rapid QC results
- Increased throughput (to enable scale-up)
- Reduced risk of radioactive spills and contamination
- Reduced personnel exposure
- No cleaning or equilibration required (disposable kit)
- Inventory reduction (tracking individual expiry dates of multiple supplies/standards)
- Process standardization across sites and/or products
Cost Reduction
- Fewer and less skilled personnel
- Faster, cheaper training
- Faster, cheaper audits
- Remote record access/auditing
- Avoid cost of addressing 483’s
- One machine to maintain
- Estimated net savings ~$26,000/year for FDG production (most common tracer)
- Estimated net savings of $56,000/year for each additional tracer beyond FDG
Size
- 34 cm (W), 98 cm (H), 67 cm (D)
- Takes up as much room as a typical GC
Scalability
- Platform for easy addition of other tracers
One instrument
- Single mode of detection for all tests
Contactless Testing
- Sample never touches the instrument
Disposable Kits
- Single use, completely disposable path
